 |
Now you may be tempted to write this
down in a first reaction equation as follows |
| |
|
|
|
|
|
|
|
|
|
 |
While this is not necessarily wrong, it is at
least strange: You create, in a kind of chemical reaction, something from
nothing - what keeps you from applying this equation to vacuum, which is surely
not sensible? Maybe you should somehow get the crystal involved as the
reference system within which things happen? |
 |
You may devise a more elaborate
system by looking at your crystal before
and after you formed a Frenkel pair. |
|
 |
Before a
Frenkel pair is formed, the site occupied by the vacancy after the formation process is a Na site, we
denote it by NaNa. This simply means that a Na atom
occupies a Na site before a vacancy is formed there. |
|
 |
At the interstitial site, where the Na
interstitial after the formation process is
going to be, you have nothing before the
process. However, all those possible interstitial sites also form a lattice
(e.g. the lattice of the
octahedral sites);
in a perfect crystal all those sites are occupied by
vacancies, we consequently denote an empty interstitial site by
Vi. |
|
 |
A Na ion on an interstitial site then is
Nai, and a vacancy becomes VNa . Now we can
write down a reaction equation that reads |
|
|
|
|
|
|
|
|
|
 |
This
looks cool, we actually create a
Frenkel pair within a crystal and not out of thin air - but don't use it uncritically with the
mass action law! We will see somewhat later why we have
to be careful here. |
|
 |
But really, the reaction
equation does look so much better this way!
Small wonder, we just invented part of the so-called Kröger-Vink notation, in use since the fifties of
the 20th century - not all that long ago, actually. |
|
 |
This notation is also called notation by structure elements and it is very useful for
formulating all kinds of reactions involving point defects. But we cannot use
them uncritically with the mass
action law, i.e. for quantitative calculations. The reason is simple, but
usually never mentioned in the context of chemical reaction formulation: |
 |
A proper reaction equation contains
only reaction partners that are
independent, i.e. you can -
at least in a thought experiment - change the concentration of either component
while keeping the concentration of all the
others constant. Consider for example the following equation: |
|
|
|
|
|
|
|
|
|
|
 |
You can, in principle, change the amount of
every reaction partner in some container
without changing the others. |
|
 |
In our equation, however, if you think about
changing, e.g. NaNa a little, you automatically change Vi, too -
those quantities are not independent! |
 |
Chapter 2.4 - often alluded to - will contain details
about all of this. It is not easy to grasp the reaction equation concept for
point defects in all its complexity, but it is worthwhile if you want to dig
deeper into point defects. For the purpose of this paragraph lets just
postulate that the two sums left and right
of the reaction equation constitute the proper reactants (they are called
building elements in the
Schottky notation) |
|
 |
The mass action law may now be applied, it
yields |
|
|
|
|
|
[NaNa + Vi]
[Nai + VNa] |
= const = exp – |
GReaction
kT |
|
|
|
|
|
|
 |
The trick, essentially, was to replace the zero
in the first equation with (NaNa
+ Vi), i.e. with a constant
representing the crystal! |
|
 |
GReaction, of course, is
the free enthalpy change of the crystal upon the formation of one Frenkel pair,
essentially HFP in our nomenclature. With
NaNa = number of lattice places and N' = number
of interstitial places, and the usual approximation that those numbers are
constant and much larger than the number of Frenkel pairs, we obtain for the
concentration cV of the Na vacancy and the
concentration ci of the Na interstitial from
the mass action law: |
|
|
|
|
|
| cV(C) · ci(C) |
= |
N'
N |
· exp – |
HFP
kT |
|
|
|
|
|
 |
Note that this is
not our old result, because cV must not be = ci
by necessity! But so far we have not worked in
electroneutrality - it was not contained
in the reaction equation. |
|
 |
Also note that just from the mass action law we
could not know that the pre-exponential factor is N'/ N.
This requires somewhat deeper thinking - we will do this in sub-chapter 2.4.3. |
 |
We might now go
through the same procedure as before by using a similar reaction equation for
Schottky defects - with a few more complications in finding the proper reaction
equation. But we will not do this here (but in the
link), just note the rather simple
result: |
|
 |
With cV(A) and
cV(C) denoting the vacancies on the anion or cation
sublattice, resp., we obtain |
|
|
|
|
|
| cV(A) · cV(C) |
= exp – |
HS
kT |
|
|
|
|
|
|
 |
with HS = formation
enthalpy of a Schottky pair. Again, this is not the old equation - the concentrations are
not necessarily equal. |
|
 |
Note that the vacancies on the anion or cation
sublattice are positively or negatively charged - opposite to the charge of the
(negatively charged) anion or (positively charged) cation that was removed! A
cation vacancy thus carries a negative charge and so on, whereas a
cation interstitial carries a positive charge. Look at the
illustrations if you are
not clear about this! |
 |
Knowing from the
direct calculation of Schottky defects
that electroneutrality has to be maintained, we introduce electroneutrality for
the general case of several charged defects, too: The sum of all charges on
point defects must be zero: |
|
|
|
|
|
|
|
|
|
|
 |
Or: Sum
over all negative charges = Sum over all positive charges.
Now we have 3 equations for 3 unknowns which can
be solved almost with ease. We obtain for
the general situation of mixed defects |
|
|
|
|
|
| |
cV(C) |
= exp – |
HS
2kT |
· |
( |
1 + |
N'
N |
· exp |
HS – HFP
kT |
) |
1/2 |
| |
cV(A) |
= exp – |
HS
2kT |
· |
( |
1 + |
N'
N |
· exp |
HS – HFP
kT |
) |
– 1/2 |
| |
ci(C) |
= |
N'
N |
· exp |
Hs
2kT |
· exp – |
HFP
kT |
· |
( |
1 + |
N'
N |
· exp |
HS – HFP
kT |
) |
– 1/2 |
|
|
|
|
|
|
 |
These equations contain the "pure"
Frenkel and Schottky case as limiting cases. Now it is time for an
exercise |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
 |
We now have the general equations and
thus can answer the essential question for this case: How likely is a mixed
case? |
|
 |
In a more quantitave fashion we ask: How
different must the formation enthalpies be if just one defect type should
dominate? |
|
 |
That is, of course, an excellent exercise
question. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
 |
If you do the exercise, you will find
out that relatively small differences in the order of 0.1 eV in
the formation energies of Schottky or Frenkel defects will already lead to the
preponderance of one defect type. |
|
 |
This is important! Since it is quite likely that
formation enthalpies for Schottky or Frenkel defects in a given crystal will
differ by more than just 0.1 eV, we are quite justified to look at the
defect situation in an "either - or" mode: "Either we will have
Frenkel defects, or Schottky defects. One kind will practically always
"win". |
 |
The final questions are then: When do
we have Schottky disorder, when do we have Frenkel disorder? What are typical
formation enthalpies? Are there general criteria for what kind is more likely
to occur in a given crystal? Can we tell or at least make an educated
guess? |
|
 |
Well let's look at a few experimental findings
(all numbers are from "Hayes and Stoneham" ; more
numbers can be found in the link): |
|
|
|
|
|
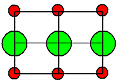
| Crystal |
Type of lattice |
Type of disorder |
Formation enthalpies HS [eV] |
<110>
projection
Ion size to scale |
Cations = red
Anions = green |
NaCl
and all other alkali halides |
NaCl-type |
Schottky |
LiF |
2,5 |
 |
| NaCl |
2,3 |
|
| KCl |
2,3 |
|
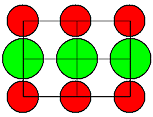
| KBr |
2,4 |
|
| CsI |
1,9 |
 |
| Some Oxides |
MgO |
5,7 |
|
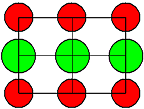
AgCl
and AgBr, |
NaCl-type
|
Frenkel
with cation interstials |
AgCl |
1,45 |
 |
| NaNO3, KNO3, .. |
complex |
|
|
|
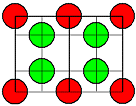
CaF2
and SrF2, BaF2 |
CaF2 type |
Frenkel with
anion interstials
("Anti-Frenkel") |
CaF2 |
2,7 |
|
| SrF2 |
2,3 |
|
| BaF2 |
1,9 |
 |
| SrCl2 |
1,7 |
|
|
|
|
|
 |
What we see is: |
|
 |
1. It is hard to make a prediction of what
kind of defect you will find. Even looking at a lattice projection with the
ions drawn to scale (using
tabulated
ionic radii), does not offer immediate clues (besides the obvious one that
you only would expect Anti-Frenkel defects
with anion interstitials, if the anion is not much larger than the
cation). |
|
 |
2. The formation enthalpies of Frenkel
defects tends to be a bit lower than those of Schottky defects. |
|
 |
3. The formation enthalpies in the more
simple ionic crystals (no oxides) tend to be rather small - around 1 eV
per single defect; quite comparable to the vacancy formation enthalpy of simple
metals. This might be taken as a hint that charge matters less than bonding and
lattice distortion. |
|
|
|
© H. Föll